Organize laboratory work in your production plant!
See how explitia.LIMS helps manage test orders, analysis results, specifications, samples, and documentation in one place. Contact us and check how it can work in your plant.
What you gain with explitia.LIMS system?
For your company, the biggest benefit is clear: the laboratory no longer works as a separate area. Instead of scattered data and manual tracking, you get one place to manage testing, quality requirements, and documentation.
If you are looking for LIMS software that supports daily work instead of only storing data, explitia.LIMS helps you organize your processes and stay in control.

How explitia.LIMS works
Test orders in one place
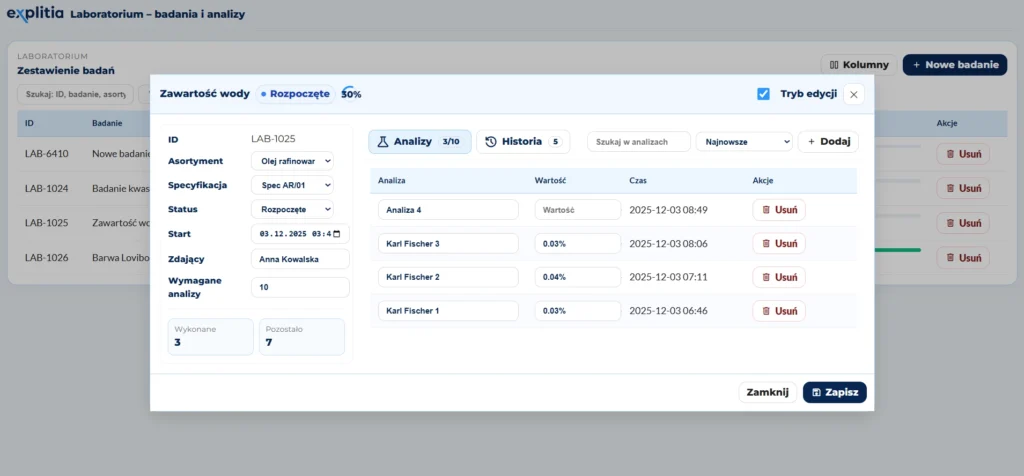
At the core of the system is the management of laboratory analysis orders. Orders can be created manually or generated automatically based on rules or testing schedules. With that, the system does more than simply store results. It also helps organize how your laboratory plans work.
Tests are no longer tracked in several places or dependent on employee memory. You know what should be done, when, and in what scope. It means better daily organization for your teams and for your company, fewer delays, fewer missed tasks, and fewer unclear situations.
Specifications and quality requirements
Specification management is an important part of a system LIMS setup. Specifications define which parameters a product, raw material, or semi-finished product must meet at a given stage of the process. These can include quality values, composition, temperature, acidity, or other properties important to your technology.
Thanks to that, you can immediately see whether a material or batch meets the required standard. It results in faster compliance assessment and shortens the path from result to decision.
Test results in process context
A test result alone is not enough if you do not know what it relates to or how it should be assessed. That is why explitia.LIMS connects analysis results with the right context: batch, process stage, quality requirements, and specification.
This makes it easier to:
- assess batch compliance
- check parameters at a specific process stage
- confirm the quality of raw materials, semi-finished products, or finished goods
- make faster and more informed operational decisions
Here, a LIMS laboratory environment displays its true value. The result is not only recorded. It is linked to quality and production decisions.
Main modules and work areas
PDF documents and certificates
Laboratories and quality departments often need to prepare documents for customers, external partners, or formal requirements. explitia.LIMS supports the creation of certificates and documents in PDF format. The document layout can be adjusted to the required format and use case.
It helps you organize sharing the results and confirmations. Instead of preparing documents manually in different places, you can build the process on data already stored in the system.

Laboratory stockroom
explitia.LIMS goes beyond test results. It can also include laboratory stockroom management, including parameters such as reagent levels, expiry dates, material availability, and usage order.
Thus, your team can stay organized, and it lowers the risk of using materials that should no longer be in use or running out of needed resources.
Calibration and sample collection scheduling
Another important area is laboratory instrument calibration planning and sample collection. The system helps you keep track of deadlines and organize activities related to measurement accuracy and regular control.
You get better oversight of what should be done and when. The laboratory works according to clear rules, and the risk of missing important actions decreases.

Samples, equipment, and resources
The solution also covers samples, equipment, and laboratory resources. A laboratory works well only when these elements are connected. Test orders, samples, parameters, documents, equipment, and materials should not function separately.
explitia.LIMS helps you manage them in one place.

Turn scattered testing and documentation into an organized process!
explitia.LIMS does more than record test results. It helps you plan analyses, assess compliance, manage documentation, and better connect the laboratory with production and quality. Write to us and see how much it can bring to your plant.
How explitia.LIMS connects with other areas
explitia.LIMS works well with other systems in the production environment, such as the Production Portal. The connection with traceability is especially natural because laboratory tests often refer to specific batches, production stages, or process events.
It also connects well with production, work orders, and SPC, because the laboratory is an important part of wider process and quality control. Integration with ERP is also possible, especially where laboratory material consumption needs to be recorded or data needs to be transferred to other parts of the organization.
Traceability
explitia.LIMS supports traceability because laboratory tests often refer to specific batches, production stages, or process events.
More
Production and work orders
The system connects with production and work orders because test results may influence decisions about releasing, continuing, or holding the process.
More
ERP
explitia.LIMS can exchange data with ERP systems, especially where laboratory material consumption needs to be recorded or data needs to be transferred to other parts of the organization.
More
SPC
The system can also work with SPC by delivering measurement and quality data needed for ongoing process stability control.
More
Business value for quality and production
A well-implemented LIMS software system helps organize laboratory work, but the value goes beyond the laboratory itself. The whole plant can benefit.
You have less scattered data because a reliable source of information about tests, results, and documents is available in one place. It is easier to assess compliance because results are compared with specifications. Teams work with more confidence because testing, sample collection, and instrument calibration follow clear rules.
There is also better visibility for quality and production. When laboratory work is connected to the actual process, it is easier to decide what should happen next with a batch, a parameter, or a product.
As a result, you can expect:
| Less manual follow-up to lab processes |
| Lower risk of errors and missed tasks |
| Better organization of laboratory work |
| Faster access to important information |
| More confidence in compliance assessment |
| Easier document preparation for customers and partners |
Who explitia.LIMS is for
This solution is for you if you want to plan tests, compare results with specifications, manage samples, and document compliance in one place.
explitia.LIMS works well in production companies where the laboratory supports quality decisions and has a direct impact on the process. It is a good choice for quality departments and laboratories that want to organize daily work and connect test results more closely with production.
How we implement explitia.LIMS
We define the scope of laboratory work and quality requirements
We organize tests, specifications, documents, and schedules
We configure modules according to the way your plant works
We connect the laboratory with quality and production processes
We expand the system with additional areas depending on your needs

Let’s talk about explitia.LIMS
Do you want to organize laboratory work and get better control over quality?
We will show you how explitia.LIMS can work in your plant, how it connects with traceability, SPC, production, and ERP, and how it helps turn laboratory data into faster and more confident decisions.